 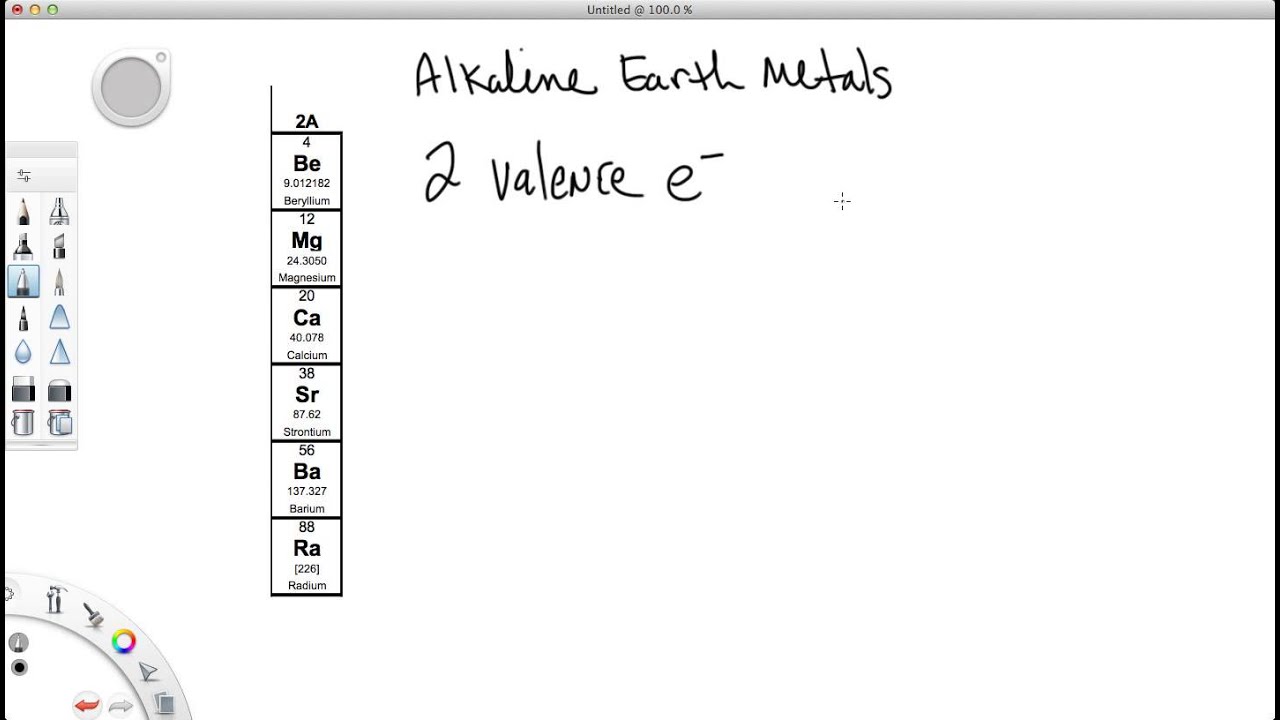
Questions Tips & Thanks Want to join the conversation Sort by: Top Voted Manahil Ahmed 9 years ago If helium is in group 8 why does it not have 8 valence electrons. To give another example, Manganese would have 7. How to determine the number of valence electrons and draw Lewis structures for main group elements starting from the electron configuration. So, Vanadium would have 5 valence electrons when including the outermost d orbital electrons. Since we are including the d electrons, we would add 3 electrons since Vanadium is in the third group of transition metals and we add one electron as we move one element to the right. Vanadium is not an exception to any rule we covered, so we can assume that its 4s orbital is completely filled (2 electrons). If a question says to include the outermost d orbital electrons, that would mean you add the amount of d orbital electrons to the 4s valence electrons. 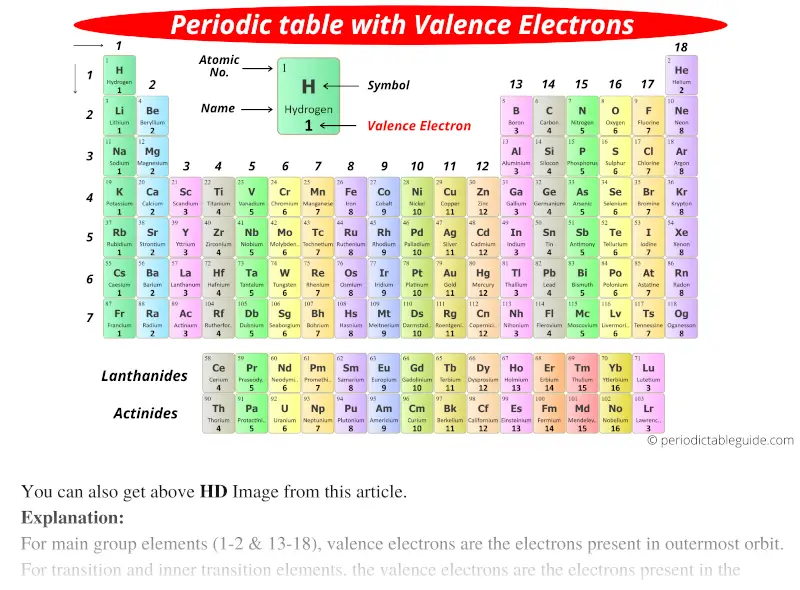
There are some exceptions to this, such as Chromium, which would have 1 valence electron due to it wanting to have 3d5 in its configuration for more stability. By finding the electron configuration of these elements you would find that they only fill the 4s orbital of the n=4 shell, and for that reason they would have 2 valence electrons (there are two valence electrons in a full 4s orbital). Point to Remember: Mostly for transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core.Since, for this class, we are only responsible for the first row of transition metals, they all have n=4 as their outermost shell. (See the above or below periodic table to see the valence electrons of inner transition metals). 
Inner transition elements can have valence electrons ranging from 3 to 16. In some inner transition metals, the electrons of incomplete d-orbitals are also considered as valence electrons. Hence, for inner transition elements, the electrons of both f-subshells as well as s-subshell behave like valence electrons. The charge depends on the atoms electron configuration and the number of valence electrons. The inner transition elements have incomplete f- subshells and they are very close to the outer s-subshell. An ionic charge is a positive or negative charge on an atom. (See the above or below periodic table to see the valence electrons of transition metals).Īlso the two bottom rows at the bottom of the periodic table are the inner transition elements (or f-block elements) also have the similar case. Hence, the transition elements (i.e d-block elements from group 3 to 12) can have more valence electrons ranging from 3 to 12. So, the electrons of both d-subshell and s-subshell behave like valence electrons. It is more difficult to find the valence electrons of transition elements as they have incompletely filled d-subshell and this d-subshell is very close to the outer s-subshell. These atoms are often called the alkali and alkaline earth elements. For example, potassium has a configuration Ar4s 1. 1 ), the configuration is a closed shell of core electrons, plus s electrons in a new shell. What about valence electrons of transition and inner transition elements?įor the transition elements and inner transition elements, the case is more complicated. For atoms found in the first two columns of the periodic table (figure 1.6.1 1.6. Well, this suits perfectly for the main group elements (i.e group 1, 2 and group 13 to 18), but what about the transition and inner transition elements? Hence, magnesium has 2 valence electrons. Here, you can see that the highest principal quantum number is 3, and the total electrons in this principal quantum number is 2. The electron configuration of magnesium is 1s 2 2s 2 2p 6 3s 2. For representative elements, the number of valence electrons equals the group number on the periodic table. The valence electrons are the electrons in the outermost shell. Lewis structures show all of the valence electrons in an atom or molecule. This also means that the number of valence electrons that an element has determines its reactivity. A Lewis structure is a way to show how atoms share electrons when they form a molecule. These electrons, being the furthest from the nucleus and thus the least tightly held by the atom, are the electrons that participate in bonds and reactions. Valence electrons can also be determined as the electrons present in the shell with highest principal quantum number (n). These electrons are most distant from the positive nucleus and, therefore, are most easily transferred between atoms in chemical reactions. Valence electrons are electrons that located in the outermost electron shell of an atom. 
The magnesium element has 2 electrons in outermost orbit. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
